Carbon nanotubes - Synthesis, Properties and Applications
- Bhavya Putta
- Jan 30, 2021
- 6 min read
Updated: Sep 21, 2024

Carbon is one of the most versatile elements that occur in nature. It is referred to as the Element of Life. Carbon forms the highest number of compounds compared to all other elements. It is largely distributed in nature. The carbon 12 isotope is the standard, relative to which the atomic masses of all other elements are measured. Carbon is the root of the evolution of the universe and also the major contributor to various technological elevations.
Carbon is the sixth element (after Hydrogen, Helium, Lithium, Beryllium, and Boron) that occurs in group 14 (P block) of the modern periodic table. Its tetravalence nature in the excited state allows it to form four covalent bonds. It has three isotopes viz carbon-12, carbon-13, and carbon-14. Carbon also has various allotropes that include Diamond, Graphite, and Fullerenes. Likewise, Carbon nanotubes are the allotropes of carbon. Their high thermal conductivity, less density, high mechanical strength, and effective shielding properties make them quite significant in Engineering and Medicine as well.
What are Carbon nanotubes?
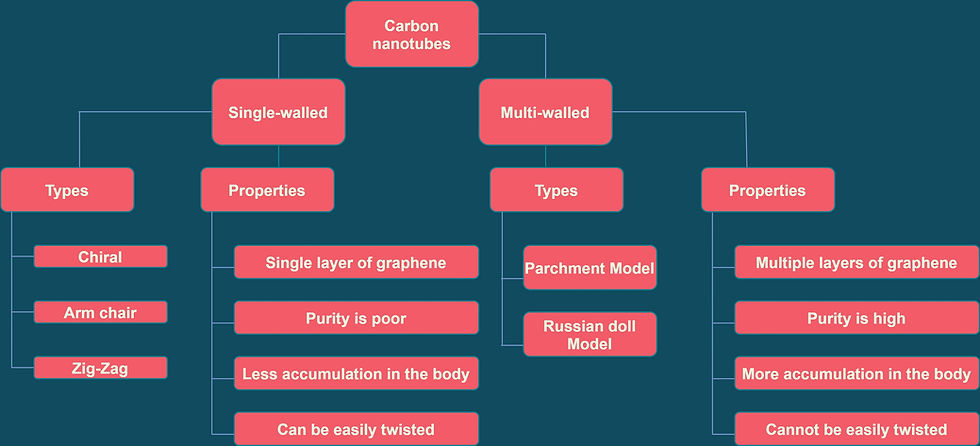
Sumio Iijima was the first one to report the discovery of nanotubes in 1991, in his ground-breaking paper Helical microtubules of graphitic carbon. Carbon nanotubes can be described as cylindrically shaped molecules that contain a single layer of carbon atoms. Graphene is an allotrope of carbon where the carbon is sp2 hybridized. Carbon nanotubes are the rolled-up structures of a single layer of graphene. They can be single-walled carbon nanotubes(SWCNTs) with a diameter less than 1 nanometer or Multi-walled carbon nanotubes(MWCNTs), consisting of several nanotubes attached concentrically with a diameter greater than 100 nanometers.

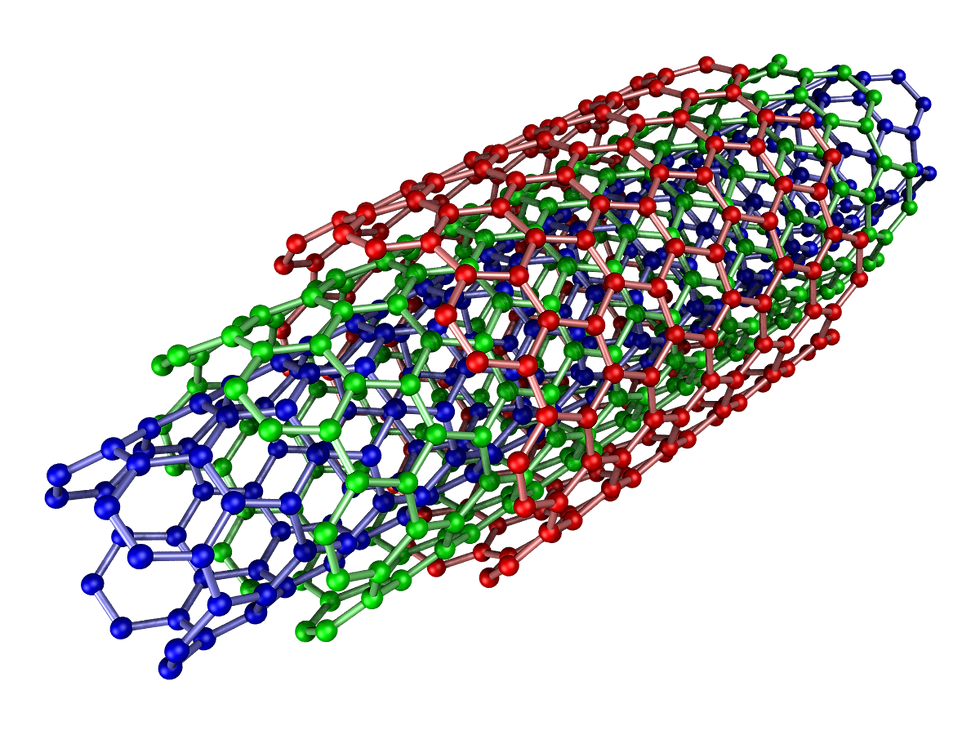
Single-walled carbon nanotubes:
They are of various types namely, armchair, chiral, and Zig-Zag.
Their structures are influenced by a pair of indices (n,m). The indices also affect the electrical properties of the nanotubes.
For m=0, the nanotubes are zigzag, and they are armchair for n=m.
The other states are chiral.
Multi-walled carbon nanotubes:
They occur in two models viz Parchment model and Russian Doll model.
Russian Doll model - A smaller nanotube is present inside a large one, and the diameter of the outer nanotube is greater than the inner one.
Parchment Model - The same single layer of graphene is wrapped around itself many times to form a multi-walled structure.
Both SWCNTs and MWCNTs have similar physical properties, but the strong intermolecular interaction and Van der Waal's forces of the MWCNTs provide them with more mechanical strength. MWCNTs are always conducting in nature, whereas the conductivity of SWCNTs depends on the chiral vector. They can behave like conductors, non-conductors, or semi-conductors.
Synthesis of Carbon nanotubes
There are currently 3 methods available for the synthesis of carbon nanotubes.
Chemical vapor deposition(CVD) technique.
Carbon arc-discharge technique.
Laser ablation technique.
Chemical vapour deposition(CVD) technique:
It is the most widely used method as large quantities of nanotubes can be produced under controllable conditions and sufficiently lower cost. It involves introducing a catalyst in a carbon involved gas reaction. This produces nanotubes of carbon on the catalyst inside a high-temperature furnace.
Initially, clusters of a secondary catalyst of the size of nanotubes are formed which turn into the nanotubes on the catalyst. The secondary catalyst decays with the formation of the carbon envelope. This process can be a catalyst(temperature up to 750 degrees Celcius) or plasma(temperatures 200-500 degrees Celcius) supported.

Carbon arc-discharge technique:
High temperatures above 1700 degrees Celcius are used in this method. Usually, arc-discharge between highly impure graphite electrodes that are placed in a chamber filled with Helium at sub-atmospheric pressure is used. The chamber consists of cathode and anode of graphite, evaporated carbon molecules, and some metal molecules viz Iron, Cobalt, etc. Direct current is passed through the chamber and is heated up to 4000K.
This results in the solidification of half of the evaporated carbon molecules onto the tip of the cathode. The remaining carbon gets deposited on the periphery and condenses to 'chamber soot' at the walls of the chamber and 'cathode soot' on the cathode. The chamber soot, cathode soot, and inner core yield single-walled and multi-walled carbon nanotubes and polyhedral graphene layers.
Laser ablation Technique:
A pure block of graphite present inside a quartz tube is heated inside a furnace at 1200 degrees Celcius using a high power laser(for example YAG type). The addition of metal catalysts to the graphite blocks is necessary. Studies have revealed that the power of the laser beam used affects the diameter of the nanotubes. The diameter of nanotubes decreases with an increase in the power of the laser beam.
This method is advantageous as the nanotubes produced are free of impurities(The metal impurities tend to evaporate once the end of the tube is closed). But it is not very economical as the synthesis requires high purity graphite blocks and high power laser beams. The nanotubes synthesized in this way are not straight and have some branching in between.
Purification methods
The nanotubes produced by the above methods are not completely pure and contain impurities. Hence purification of end products is necessary at the end of each process. The various methods used for the purification of nanotubes are listed below.
Treating the nanotubes with acids
Studies have shown that boiling of SWCNTs in a solution of Nitric acid or Hydrochloric acid effectively removes any amorphous impurities.
Sonication
After the SWCNTs are oxidized and treated with acids, the carbon tubules present inside them are removed by the sonication process.
Other methods include:
Using Size-exclusion Chromatography.
Blending of DNA-SWNT and ion-exchange chromatography.
Fluorination and Bromination processes.

Properties
Usually, they have a resistivity of 10^-4 ohm-m.
The maximum current density can be 1,013 A/m^2.
Density varies from 1.33 g/cm^3 to 1.4 g/cm^3 depending on the indices (n,m).
The thermal conductivity is approximately 2,000 W/m/K. They can withstand very high temperatures.
The relaxation time is about 10 to 11 seconds.
An interesting property of the nanotubes is Elasticity. Their maximum tensile strength is 100 Gpa. The deformation due to a high force is lost on the removal of the force, and the nanotube acquires its original structure. But this elasticity property is lost when an extremely powerful force is applied.
The youngs modulus of SWCNTs is around 1 TPa and that of the MWCNTs is 1.28 TPa.
Applications
Carbon nanotubes have many amazing applications due to their properties of elasticity, effective shielding, and high thermal conductivity.
Sensors - Though the nanotubes are sturdy and inert, studies have shown that they are extremely sensitive to chemical doping and charge transfer. Thus CNTs are effective for developing the FETs(Field effect transistors). They are used in identifying the greenhouse gases from pollutants.
Transistors - Despite the development of many useful materials(such as graphite), the nanotubes are considered strong ones for the next generation of ultra-fast and thin transistors. Researchers believe that the nanotubes exhibit excellent transistor behaviour even in the nanometer range.
Biomedical Applications - The nanotubes find exclusive applications in the biomedical industry. They are used in cancer cell recognition, Tissue engineering, Artificial implants, Drug manufacture, and delivery.
Catalysts - The large surface area of nanotubes and their ability to attach with other chemical species easily make them excellent catalysts. They are currently used as catalysts in many chemical reactions. Controlling their catalytic activity is quite difficult.
Filtration - The carbon nanotubes are prominent for the filtration of water efficiently. Scientists have demonstrated that the nanotubes fixed in fatty membranes compress the inflow water molecules into a single chain and thus promote faster transport. They also protect from various biological agents such as bacteria and viruses due to their extremely small pores.
Apart from these, the nanotubes are also used in Bucky papers, Nano inks, Displays, Electrodes, Synthetics, Photonic and Optoelectronic applications.
Conclusion
A few barriers in using CNTs are their insolubility in aqueous media, toxicity, and biodistribution. Though some issues need to be resolved, their homogeneity, small diameters, and effective encapsulation of other materials inside them make them very popular. The nanotubes find many potential applications in medicine. Their physical and chemical properties promote regenerative medicine. They can also be adopted to design ultra-thin, high-speed devices. If used effectively, the carbon nanotubes promise a splendid future in Engineering and medicine as well.
See Also
Order Electronics Projects
Want us to guide you through your project or make the project for you? Click on the button below or reach out to us via Call/WhatsApp at (+91) - 7600948607
You can -
Order Basic Electronics Projects
Order Embedded Systems Projects
Order IoT Projects
Order FPGA Projects
Order VLSI Projects
Order Image Processing Projects
Order Matlab Projects
Order TinkerCAD Projects
Order Proteus Projects
Click on the button below to fill out the project inquiry form -
Create Various Projects
Check out our Free Arduino Projects Playlist - Arduino Projects
Check out our Free Raspberry Pi Projects Playlist - Raspberry Pi Projects
Check out our Free TinkerCAD Projects Playlist - TinkerCAD Projects
Check out our Free IoT Projects Playlist - IoT Projects
Check out our Free Home Automation Projects Playlist - Home Automation Projects
Check out our Free NodeMCu Projects Playlist - NodeMCu Projects
Follow us -
Please do follow us i.e. #learnelectronicsindia to get daily updates about new blogs, videos, courses, products, offers, competitions, quizzes, and Internship Opportunities.










LearnElectronics India's explanation is spot-on. Great work!
Hats off to Learn Electronics India for producing an outstanding blog on Carbon Nanotubes! This article strikes a perfect balance between technical information and readability. The way it explained the synthesis techniques and the wide range of properties exhibited by Carbon Nanotubes was impressive. Moreover, the insights into their practical applications in electronics and other industries were eye-opening.